Revolutionizing Neuroscience: MIT Spinout Develops Hair-Thin Fibers for Brain Integration
Starting a company often requires a bold vision, and NeuroBionics exemplifies this daring spirit. This MIT spinout aims to revolutionize the treatment of neurological disorders such as depression, epilepsy, and Parkinson’s disease, potentially improving the lives of millions.
Innovative Technology for Neurological Disorders
NeuroBionics is making waves in the medical technology space with its innovative approach to neuromodulation therapy. According to renowned investor Steve Jurvetson from Future Ventures, this 18-month-old startup could significantly impact the peripheral nervous system, addressing issues related to pain, incontinence, and beyond.
How NeuroBionics Works
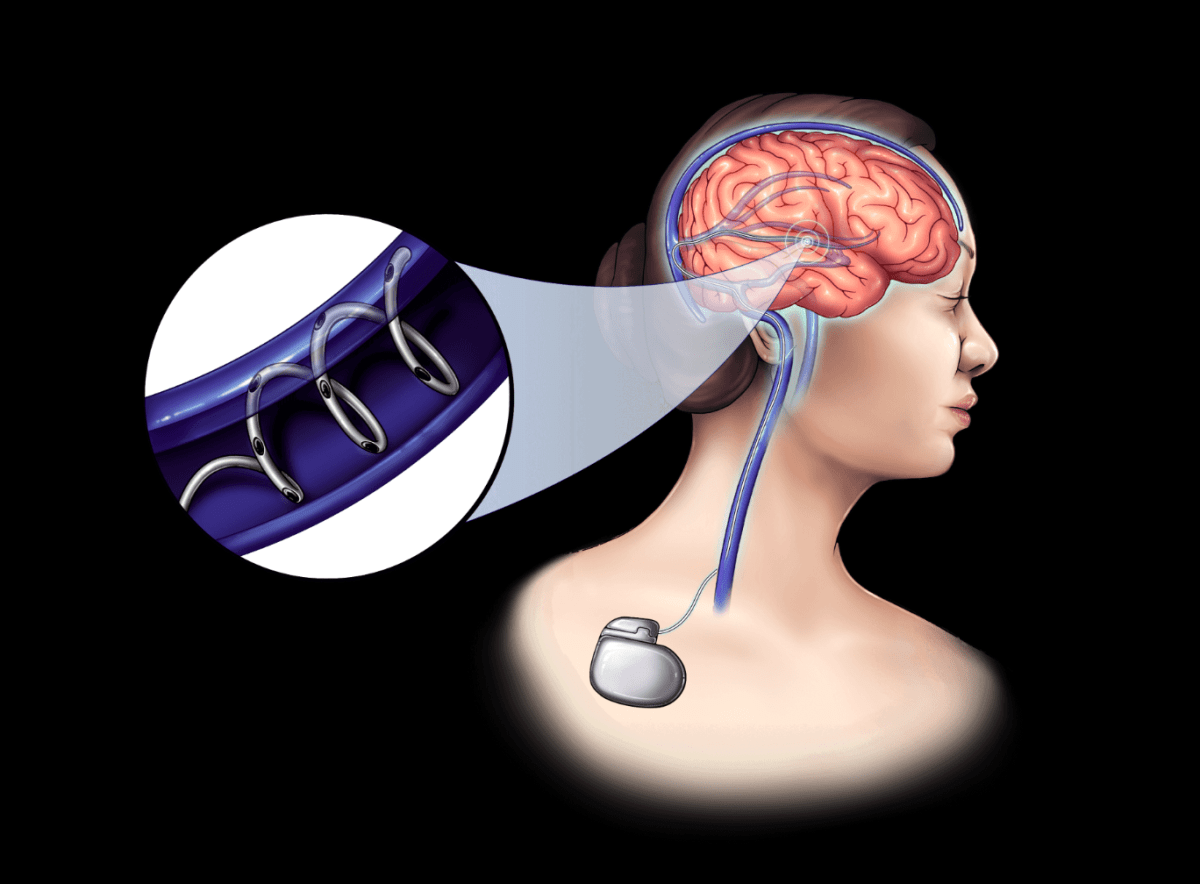
So, how does NeuroBionics plan to achieve its ambitious goals? The company is developing bioelectric fibers that are as thin as a human hair. These fibers are designed to be inserted into the brain’s blood vessels using a stent-like procedure, offering an alternative to traditional deep brain stimulation.
- Minimally Invasive: Unlike conventional methods that require drilling into the skull, NeuroBionics’ technique is less invasive.
- Long-lasting Power: The device is powered by an implantable battery resembling an AirPod case, which can last between five to ten years.
- Advanced Materials: Instead of using metals that can degrade and cause tissue damage, NeuroBionics employs carbon nanotubes, which are more durable and compatible with MRI scans.
The Journey of NeuroBionics’ Founders
The inception of NeuroBionics stems from over a decade of research at MIT. MJ Antonini, the CEO, co-founded the startup while completing his studies. He holds dual doctorate degrees from both Harvard and MIT through a specialized program in Health Sciences and Technology.
A Unique Educational Path
Antonini’s journey involved:
- Two years in medical school at Harvard.
- Extensive studies in medical engineering and medical physics at MIT.
- Transitioning from research to entrepreneurship to create a viable medical product.
After his academic tenure, Antonini collaborated with fellow researcher Nicki Driscoll, who now serves as the CTO of NeuroBionics. Together, they aim to bring their groundbreaking technology to market.
Future Prospects and Funding
The road ahead for NeuroBionics is filled with potential yet requires substantial groundwork. Recently, the startup secured $5 million in funding from Dolby Family Ventures, Future Ventures, GreyMatter Capital, and additional investors. This capital will help them refine their clinical device.
Next, NeuroBionics plans to conduct safety and efficacy trials in pigs, as these animals share crucial anatomical and physiological similarities with humans. Following these trials, they will submit their findings to the FDA for review and potentially apply for an investigational device exemption (IDE).
Projected Market Entry
When asked about the timeline for bringing their technology to market, Antonini suggested a target year of 2030. This timeline reflects the complex nature of medical device development and regulatory approval.
Transforming Medical Treatment
NeuroBionics’ technology has the potential to open doors for patients who currently avoid deep brain stimulation due to its invasive nature. Jurvetson emphasizes that traditional treatments are often declined by those who could benefit, as they require significant surgical intervention.
With NeuroBionics’ minimally invasive stent technology, the landscape of neurological treatment could change dramatically, offering new hope to many patients in need.
For more information on innovative medical technologies, visit our Medical Innovation page.